
A team of scientists from Andalusia has studied the effect of radiation on cancer stem cells (CSCs) and the CSC microenvironment in both in vitro and in vivo models. The study showed that this effect can be determined from patients’ blood samples, which would be very useful for developing more personalized precision treatments for women with breast cancer undergoing radiotherapy.
The results have been published in three prestigious scientific journals: Cancers; Molecular Oncology; and Journal of Cellular and Molecular Medicine
Breast cancer is the most common cancer among women and one of the leading causes of cancer death worldwide. The presence of cancer stem cells (CSCs) in tumours is associated with a higher rate of relapse, metastasis, and resistance to treatment with chemotherapy and radiotherapy. Therefore, the radiotherapy (RT) that is so extensively used for the treatment of breast cancer is not always effective, as it is the heterogeneity and biological complexity of the tumour (and, mainly, the CSCs that are part of it) that are responsible for the recurrence of cancer and metastasis following RT.
The microenvironment or niche that surrounds the tumour is another important factor to consider when determining the efficacy of RT among women with different molecular types of breast cancer. Matrix metalloproteinases (MMPs) are enzymes involved mainly in the remodelling of the extracellular matrix that forms the tumour niche, and they are also involved in carcinogenic processes such as inflammation, the formation of new vessels (angiogenesis), invasion, and metastasis.
It was for this reason that this team of scientists conducted two separate studies to demonstrate that small gene-regulatory molecules (microRNAs) specific to CSCs and MMPs related to the CSC microenvironment are key to predicting the breast cancer patients’ response to RT treatment, depending on the molecular type and the radiation dose used. The studies were led by Professor Juan Antonio Marchal Corrales of the Department of Human Anatomy and Embryology of the UGR, Director of the Advanced Therapies: Differentiation, Regeneration and Cancer Research Group (CTS-963), and by Professor María Isabel Núñez Torres of the UGR’s Department of Radiology and Physical Medicine, member of the Basic and Clinical Oncology Research Group (CTS-206). As well as their roles at the UGR, both researchers also belong to the Biohealth Research Institute in Granada (ibs.GRANADA) and the Biopathology and Regenerative Medicine Institute (IBIMER).
These studies are the fruit of a multidisciplinary project in which translational researchers, bioinformatics experts, and clinical researchers from the Universities of Granada and Jaén and the Virgen de las Nieves University Hospital (Granada)—all of which are members of ibs.GRANADA and the Andalusian Network for the Design and Translation of Advanced Therapies—have joined forces to make further advances in the fields of Personalized Medicine and Precision Medicine in Oncology.
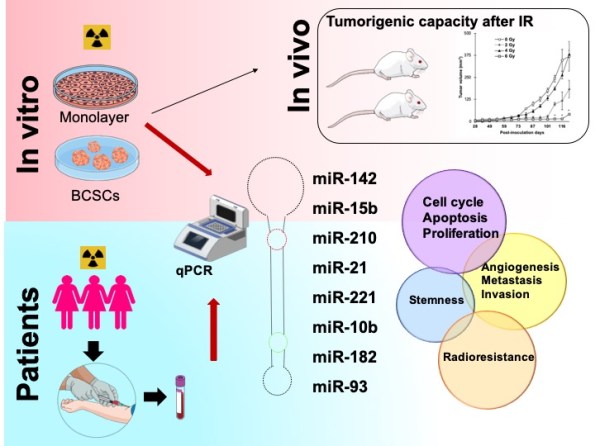
In these studies, it was demonstrated that exposure to radiation doses of 2, 4, or 6 Gy affects the phenotype and functional characteristics of CSCs. It was also shown that the microenvironment plays a crucial role in terms of cellular interactions and CSC plasticity in tumour growth following RT.
The tumorigenic capacity of human triple-negative breast cancer CSCs previously treated with the different doses of radiation—following their orthotopic transplantation (implantation) into the mammary tissue of female mice—was evaluated. The results showed an increase in the tumour volume of the cells exposed to low radiation doses (2 and 4 Gy) compared to both those treated with high doses (6 Gy) and the controls not treated with radiation.
This would support the hypothesis that low doses of radiation in triple-negative breast cancer are insufficient to eradicate CSCs, due to the radioresistance they present; furthermore, these doses might even contribute to tumour growth. Indeed, conventional RT has been extensively used in the treatment of breast cancer (2 Gy/fraction daily; total 45–50 Gy), but hypofractionated RT is increasingly used, which involves delivering higher doses of radiation combined with shorter treatment times. The results of this experimental study suggest, therefore, that in these cases the hypofractionated RT would be the most appropriate to avoid the selection of more resistant and aggressive CSCs.
In recent years, it has been shown that variations in the expression of miRNAs and MMPs can contribute to the development of breast cancer in different molecular subtypes following different radiation doses. Therefore, these researchers studied the expression of eight micro RNAs, also known as miRNAs (miR-210, miR-10b, miR-182, miR-142, miR-221, miR-21, miR-93, and miR-15b). These play an important role both in carcinogenesis and in maintaining the characteristics of the CSCs and radioresistance. These miRNAs presented variability of expression according to the molecular type of the breast cancer under study and the different subpopulations of tumour cells post-irradiation. For example, it was shown that miR-21 and miR-182, which are associated with the response to radio-induced DNA damage, increased their expression levels after CSC irradiation for both triple-negative and hormone-dependent breast cancer, which is linked to their increased radioresistance. In contrast, the expression of mi-142 and miR-15b (associated with increased sensitivity to radiation) increased after treatment with high doses of radiation in hormone-dependent breast cancer CSCs. Therefore, these miRNAs linked to radioresistance, tumour growth, and radiosensitivity could be used as radio-response biomarkers in breast cancer.
Similarly, the study also focused on MMPs, which are enzymes regulated by their endogenous inhibitors (tissue inhibitors of metalloproteinases, or TIMPs) and by epigenetic mechanisms by histone deacetylases (HDACs). The results showed that, with radiation, the expression levels of MMP-1 and MMP-3 increased in triple-negative breast cancer CSCs, which points to their importance in the process of invasion and metastasis post-treatment.
Finally, those same miRNAs and MMPs were analysed in breast cancer patients before, during, and after RT. Variation in the expression levels of these molecules was observed throughout the RT, as was a statistically-significant relationship between these levels and certain variables linked to the particular characteristics of the patient and the tumour biology (such as the histological grade of tumour, age, condition, menopausal state, treatment with chemotherapy or toxicity). Furthermore, among patients with tumour recurrence, an increase in the blood levels of all the MMPs analysed and a decrease in TIMP-1 levels were found. Likewise, statistically-significant relationships were found for MPMP-3, MMP-9, TIMP-3 and TIMP-4, as well as in miR-182, miR-21, miR-142, miR-210, and miR-10b, with some variables analysed. This study points to the use of these molecules as possible prognostic and predictive biomarkers of RT-response and radiotoxicity in patients with breast cancer.
In light of these studies, the researchers conclude that the clinical implementation by liquid biopsy of certain miRNA and MMP signatures could be very useful in determining the efficacy of RT and in achieving personalized, precision RT dosage regimens. This would improve the prognosis, treatment, and survival of patients with breast cancer.
The study was financed by the Instituto de Salud Carlos III thanks to the Integrated Project of Excellence for Accredited Institutes (PIE16-00045), the Andalusian Department of Health, and the “Doctores Galera y Requena” Chair in Research on Cancer Stem Cells at the UGR.
Bibliography:
- Griñán-Lisón C, Olivares-Urbano MA, Jiménez G, López-Ruiz E, Del Val C, Morata-Tarifa C, Entrena JM, González-Ramírez AR, Boulaiz H, Zurita Herrera M, Núñez MI*, and Marchal JA* (2020). ‘miRNAs as radio-response biomarkers for breast cancer stem cells’, Mol Oncol 14: 556–570. doi: 10.1002/1878-0261.12635.
- Olivares-Urbano MA, Griñán-Lisón C, Zurita M, Del Moral R, Ríos-Arrabal S, Artacho-Cordón F, Arrebola JP, González AR, León J, Marchal JA*, and Núñez MI* (2020). ‘Matrix metalloproteases and TIMPs as prognostic biomarkers in breast cancer patients treated with radiotherapy: A pilot study’, J Cell Mol Med 24(1):139–148. doi: 10.1111/jcmm.14671.
- Olivares-Urbano MA, Griñán-Lisón C, Ríos-Arrabal S, Artacho-Cordón F, Torralbo AI, López-Ruiz E, Marchal JA*, and Núñez MI* (2019). ‘Radiation and stemness phenotype may influence individual breast cancer outcomes: The crucial role of MMPs and Microenvironment’, Cancers 11(11). pii: E1781. doi: 10.3390/cancers11111781.
Image captions:
Media enquiries:
Juan Antonio Marchal Corrales
Department of Human Anatomy and Embryology
Faculty of Medicine, University of Granada
Tel.: +34 958 241000 Ext. 20080 / 958 249321
Email: jmarchal@ugr.es
María Isabel Núñez Torres
Department of Radiology and Physical Medicine
Faculty of Medicine, University of Granada
Email: isabeln@ugr.es



