Niceto R. Luque Sola
Ph.D in Bioinspired Control Systems (R&D)

about me
Who am I?

My name is Niceto Rafael Luque and I am currently an assitant professor and researcher at the CICIT, University of Granada and also an associate researcher of the Institut de la Vision. University Sorbonne
I received my B.S in Electronics Engineering and a M.S. in Automatics and Industrial Electronics from the University of Cordoba (Spain) in 2003 and 2006, respectively. In April 2007 I officially joined to the University of Granada with a National Grant as a researcher of the European Project SENSOPAC .I also received my M.S. in Computer Architecture and Networks from the University of Granada in 2007.
Finally, I received my Doctorate from the University of Granada in 2013 in Control Engineering and Computer Science.
From 2012 to 2014, I participated in an EU project related to adaptive learning mechanisms and bio-inspired control REALNET. In August 2014, I officially joined the Human Brain Project (HBP); a ten-year, large-scale European research initiative whose goal is to better understand the human brain and its diseases and ultimately to emulate its computational capabilities. In 2015, I obtained an IF Marie Curie Post-Doc Fellowship from the EU Commission, I moved to Dr. Arleo's lab in Paris to work in cerebellar ageing. In 2018, I obtained a Juan de la Cierva Incorporation Post-Doc Fellowship from the Spanish Government, I moved to Prof. Ros' lab to study cerebellar adaptation and its involvements in ageing.Finally, in 2020 I got a possition at UGR as assitant professor. My main research interests include biologically processing control schemes, light weight robots, and cerebellar spiking neural networks.
Education
- University Cordoba / Bachelor's Degree in Industrial Electronics. Electronic Engineering, Power Electronics, Industrial Programing, Analog PCB Hardware Modeling Engineer in Industrial Electronics
- 2000-2004
- University Cordoba / Degree in Automatic Engineering and Industrial Electronics. Control Engineering, Industrial Robotics, Modeling Dynamical Systems and Automatisation
- 2004-2006
- University Granada / Official Master in Computer Architecture and Networks (it received a quality award from the Spanish Ministry of Education) Computer Architecture and Networks with specialization in Neuromorphic Engineering, Robotics and Neural Networks.
- 2006-2007
- University Granada / PhD,Bio-inspired Robotic Control Schemes Using Biologically Plausible Neural Structures
- 2007-2013
Expertise
- Peninsular del Laton / Practices at Cordoba (Spain) in the electronic/electrical maintenance team at Peninsular del Laton
- 2004
- CIDESUR. / Electrical engineer, Maintenance, assembly and legalization of elevators/service lifts located in Granada (Spain)
- 2005-2006
- Sevensolutions / Engineer in PCB develop and design Developing and creating PCBs, analog design, integration with ICPs
- 2006
- University of Granada / Pre-Doc Researcher in the field of neurobotics.Grant from the Ministry of Education's University Faculty Training (FPU) programme.
- 2007-2013
- University of Granada / Post Doc. Researcher in the field of neurobotics
- 2013-2015
- University Sorbonne,Institute of Vision / Marie-curie Post-Doc Fellow
- 2015-2017
- University of Granada, Prof. Ros' lab / Juan de la Cierva Post-Doc Fellow
- 2018-2020
- University of Granada,Dept.Computer technology and architecture /Assistant professor
- 2020-present

My work
Experimental studies about the Central Nervous System (CNS) in all levels (sub cellular, cellular and at system level) are performed in order to obtain a better understanding of its anatomic structures and the physiological processes that the CNS seems to possess. Nevertheless, the observations to be done with that aim must be managed within a representative scenario where the functional description of the CNS is available. This is possible just in case when all the needed conceptual elements that properly describe the CNS functionality are available too. Both Physiologists and Neurophysiologists have traditionally used the performance (or the lack of performance in presence of pathologies), as the basis for the functional assessment of the CNS components, thus producing useful qualitative and phenomenal models. Although these models are often more than enough for clinical issues they do not provide a detailed comprehension of the whole CNS.
The current technology allows a restricted in vivo access to the CNS (mainly to the more external areas) by means of functional magnetic resonance imaging and magnetoencephalography. Similarly, it is of common use, recordings by means of electrode matrices; however these recordings just allow extracellular access of barely a hundred neurons at best.
But most of the functional neural networks related to the hippocampus and the cerebellum (two of the best-known regions) are sized from just a hundred of thousand to several millions of cells. The information process within these neural networks occurs thanks to the self-organized dynamic patterns of the neural activity that covers a large proportion of the nervous system. These emerging patterns can be hardly understood taking into account just individual activities of individual cells ( or even hundred of cells) in the same way that it is tough to understand a book just taken into account individual words. Even the data collected from very large scale studies do not present the necessary resolution for observing these patterns and making the corresponding cellular interaction matches.
The biologically plausible computational models (cerebellum, inferior olive nucleus, cuneate nucleus ...etc) give answer to this problem allowing the study of neural network models " as large as it is needed" using neuronal models that have been developed according to experimental cellular data. These neural network models can be computationally simulated in pretty different conditions and circumstances to give a pretty consistent idea about how the CNS neural networks may operate. In many cases, these models are becoming a fundamental tool in the neuroscience hypothesis-experimentation cycle. The computational models allow researchers to test their "what's up when ...?" in simulation. This fact leads to make better hypothesis and better experiments designed with greater probability of success.
From this perspective, and thanks to the exponential computational performance evolution, Computational Neuroscience has positioned over the last years as a promising sub-field in neuroscience. Computational Neuroscience must not be considered as just a tool to better understand the behavior of a functional neural network within the CNS by using a mathematical analysis and massive computational simulations but also as a fundamental element to determine 1) what the different parts of the CNS do 2) How these different parts do what they do
In such scenario I have been developing my research during these years in the framework of two European projects (SENSOPAC and REALNET) helping to develop different models of diverse nervous system elements(cerebellum, inferior olive nucleus and cuneate nucleus) in cooperation with different research groups from neurophysiology(Egidio D'Angelo) trough computational neurobiology(Angelo Arleo) to robotics(Patrick van der Smagt).
Download Code
Research
- SPIKEFORCE "Real-time Spiking Networks for Robot Control" ", EU-project. From: 5/1/2002 to 4/30/2005
- SENSOPAC "SENSOrimotor structuring of Perception and Action for emergent Cognition", EU-project. From 01/01/2006 to 31/12/2009
- REALNET "Realistic Real-time Networks: computation dynamics in the cerebellum, EU-project". From 01/02/2011 to 31/01/2014
- HBP "HUMAN BRAIN PROYECT, EU-project, Horizon 2020, FET Flagship Programme". From 2013 to 2023
- SPIKECONTROL "SPIKE CONTROL, H2020-MSCA-IF-2014 EU-project". From 2015 to 2017
-
Download Poster Sensopac
-
Download Poster SensoPac 1
-
Download Poster SensoPac 2
-
Download Poster Sensopac 3
-
Download Poster Realnet
-
Download Thesis
-
Download Thesis Presentation prezi
Thesis
Journal Papers
- N. R. Luque, F. Naveros, I. Abadía, E. Ros, and A. Arleo "Electrical coupling regulated by GABAergic nucleo-olivary afferent fibres facilitates cerebellar sensory-motor adaptation", Neural Networks in press. 2022. doi: 10.1016/j.neunet.2022.08.020
- N. R. Luque*, F. Naveros*, D. Sheynikhovich, E. Ros, and A. Arleo "Computational epidemiology study of homeostatic compensation during sensorimotor aging", Neural Networks in press. 2021.* These authors equally contributed doi: 10.1016/j.neunet.2021.11.024
- I. Abadía*, F. Naveros*, E. Ros, R. R. Carrillo, and N. R. Luque "A cerebellar-based solution to the nondeterministic time delay problem in robotic control", Science Robotics, vol.6, issue 58. 2021.* These authors equally contributed doi: 10.1126/scirobotics.abf2756
- I. Abadía*, F. Naveros*, J. A. Garrido ,E. Ros and N. R. Luque "On robot compliance. A cerebellar control approach", IEEE T Cybernetics, (in press). 2020.* These authors equally contributed doi: 10.1109/TCYB.2019.2945498
- F. Naveros*, N. R. Luque *, E. Ros and A. Arleo "VOR Adaptation on a Humanoid iCub Robot using a Spiking Cerebellar Model.", IEEE T Cybernetics, 2020* These authors equally contributed doi: 10.1109/TCYB.2019.2899246
- N. R. Luque , F. Naveros, R. R. Carrillo,E. Ros and A. Arleo "Spike burst-pause dynamics of Purkinje cells regulate sensorimotor adaptation", Plos Computational Biology. 15(3): e1006298, 2019. doi: 10.1371/journal.pcbi.1006298
- R. R. Carrillo, F. Naveros, E. Ros and N. R. Luque "A Metric for Evaluating Neural Input Representation in Supervised Learning Networks", Frontiers in Neuroscience, Systems Biology 2018. doi: 10.3389/fnins.2018.00913
- F. Naveros, J. A. Garrido, R. R. Carrillo, E. Ros and N. R. Luque "Event- and Time-Driven Techniques Using Parallel CPU-GPU Co-Processing for Spiking Neural Networks", Frontiers in Neuroinformatics 2017. doi: 10.3389/fninf.2017.00007
- E. D’Angelo, A. Antonietti, S. Casali, C. Casellato, J. A. Garrido, N. R. Luque, L. Mapelli, S. Masoli, A. Pedrocchi, F. Prestori, M. F. Rizza, E. Ros. "Modeling the Cerebellar Microcircuit: New Strategies for a Long-Standing Issue", Frontiers in Cellular Neuronscience 2016. doi: 10.3389/fncel.2016.00176
- J.A. Garrido, N. R. Luque ,S. Tolu and E. D Angelo. "Oscillation-driven Spike-timing Dependent Plasticity Allows Multiple Overlapping Pattern Recognition in Inhibitory Interneuron Networks", International Journal of Neural Systems 2016. doi: 10.1142/S0129065716500209
- N. R. Luque* ,J.A. Garrido*, F. Naveros, R.R.Carrillo, E. D Angelo and E. Ros. "Distributed Cerebellar Motor Learning; a Spike-Timing-Dependent Plasticity Model", Frontiers in Computational Neuronscience 2016.*These authors contributed equally to this work doi: 10.3389/fncom.2016.00017
- A. Antonietti, C. Casellato ,J.A. Garrido, N. R. Luque, F. Naveros, E. Ros, E. D Angelo and A. Pedrocchi. "Spiking neural network with distributed plasticity reproduces cerebellar learning in eye blink conditioning Paradigms ",Biomedical Engineering, IEEE Transactions on 2015. doi: 10.1109/TBME.2015.2485301
- E. D‘Angelo, L. Mapelli, C. Casellato, J. A. Garrido, N. R. Luque, J. Monaco, F. Prestori, A. Pedrocchi and E. Ros. "Distributed circuit plasticity: new clues for the cerebellar mechanisms of learning", The Cerebellum.2015. doi: 10.1007/s12311-015-0711-7
- N. R. Luque*, J.A. Garrido*, R.R. Carrillo, E. D'Angelo* and E. Ros*. "Fast convergence of learning requires plasticity between inferior olive and deep cerebellar nuclei in a manipulation task: a closed-loop robotic simulation ", Frontiers in Computational Neuroscience.2014.*These authors contributed equally to this work doi: 10.3389/fncom.2014.00097
- C. Casellato, A. Antonietti, J. A. Garrido, R. R. Carrillo, N. R. Luque, E. Ros, A. Pedrocchi, E. D'Angelo. "Adaptive Robotic Control Driven by a Versatile Spiking Cerebellar Network ", PLOS ONE .2014. doi: 10.1371/journal.pone.0112265
- N. R. Luque*, R.R. Carrillo *, F. Naveros, J.A. Garrido, M.J. Sáez-Lara." Integrated neural and robotic simulations. Simulation of cerebellar neurobiological substrate for an object-oriented dynamic model abstraction process", Robotics and Autonomous Systems.2014.*These authors contributed equally to this work doi: 10.1016/j.robot.2014.08.002
- F. Naveros, N. R. Luque,J.A. Garrido, R.R. Carrillo, M. Anguita and E. Ros. "A spiking neural simulator integrating event-driven and time-driven computation schemes using parallel CPU-GPU co-processing. A case study", IEEE Transactions on Neural Networks and Learning Systems.2014 doi: 10.1109/TNNLS.2014.2345844
- J.A. Garrido*,N. R. Luque*, E. D'Angelo and E. Ros. "Distributed cerebellar plasticity implements adaptable gain control in a manipulation task: a closed-loop robotic simulation", Frontiers in Neural Circuits 7:159. 2013.*These authors contributed equally to this work doi: 10.3389/fncir.2013.00159
- J-B.Passot,N. R. Luque and A. Arleo. "Coupling internal cerebellar models enhances online adaptation and supports online consolidation in sensorimotor tasks", Frontiers in Computational Neuroscience 7:95., 2013. doi: 10.3389/fncom.2013.00095
- S. Tolu, M. Vanegas, J.A. Garrido, N. R. Luque, and E. Ros. "Adaptive and Predictive Control of a Simulated Robot Arm",Int. Journal of Neural Systems,23(2), pp. 0-15, 2013. doi: 10.1142/S012906571350010X
- N. R. Luque, J.A. Garrido, J. Ralli, J.J Laredo, and E. Ros, "From Sensors to Spikes: Evolving Receptive Fields to Enhance Sensori-Motor Information in a Robot-Arm",Int. Journal of Neural Systems, 22(4),1pp. 1-20, 2012. doi: 10.1142/S012906571250013X
- S. Tolu, M. Vanegas, N. R. Luque, J.A. Garrido and E. Ros. "Bio-inspired Adaptive Feedback Error Learning Architecture For Motor Control",Biological Cybernetics, 106(8-9), pp. 507-522, 2012. doi: 10.1007/s00422-012-0515-5
- N. R. Luque, J. A. Garrido, R. R. Carrillo, O. J. M. D. Coenen, E. Ros, Cerebellar Input Configuration Toward Object Model Abstraction in Manipulation Tasks. IEEE Transaction on Neural Networks, 22(8), pp. 1321-1328, 2011. doi: 10.1109/TNN.2011.2156809
- N. R. Luque, J. A. Garrido, R. R. Carrillo, S. Tolu, E. Ros, Adaptive Cerebellar Spiking Model Embedded In The Control Loop: Context Switching And Robustness Against Noise, Int. Journal of Neural Systems, 21(5), pp. 385-401, 2011. doi:10.1142/S0129065711002900.
- N. R. Luque, J. A. Garrido, R. R. Carrillo, O. J. M. D. Coenen, E. Ros, Cerebellarlike Corrective Model Inference Engine for Manipulation Tasks, IEEE Transactions on Systems, Man, and Cybernetics, Part B: Cybernetics, 41(5), 2011 doi: 10.1109/TSMCB.2011.2138693.
- J. A. Garrido, N. R. Luque, E. Ros,E. D'Angelo. "Enhancing Learning Precision At Parallel Fiber - Purkinje Cell Connections Through Deep Cerebellar Nuclei Ltd And Ltp" FENS 6 pp. 153.04, (2012). In 8th FENS Forum of Neuroscience, Barcelona,Spain, 2012.
- C. Casellato, A. Pedrocchi, J.A. Garrido, N. R. Luque, G. Ferrigno, E. D'Angelo, E. Ros."A unified motor control loop of a human-like robotic arm: feedforward, feedback and cerebellum-based learning",In Biomedical Robotics and Biomechatronics (BioRob), 2012 4th IEEE RAS & EMBS International Conference on pp. 562-567 (2012). doi: 10.1109/BioRob.2012.6290791.
- N. R. Luque, J. A. Garrido, R. R. Carrillo, E. Ros, Context Separability Mediated by the Granular Layer in a Spiking Cerebellum Model for Robot Control. International Work-Conference on Artificial Neural Networks (IWANN 2011). Advances in Computational Intelligence. Lecture Notes in Computer Science, 6691, pp. 537-546. Springer, Heidelberg (2011). doi:10.1007/978-3-642-21501-8_67.
- J. A. Garrido, R. R. Carrillo, N. R. Luque, E. Ros, Event and time driven hybrid simulation of spiking neural networks. International Work-Conference on Artificial Neural Networks (IWANN 2011). Advances in Computational Intelligence. Lecture Notes in Computer Science, 6691, pp. 554-561. Springer, Heidelberg (2011). doi:10.1007/978-3-642-21501-8_69.
- N. R. Luque, J. A. Garrido, R. R. Carrillo, E. Ros, Cerebellar spiking engine: Towards object model abstraction in manipulation. International Joint Conference on Neural Networks (IJCNN 2010). doi: 10.1109/IJCNN.2010.5596531.
- J.-B Passot.; N. R. Luque, and Arleo, A. Internal models in the cerebellum: a coupling scheme for online and offline learning in procedural tasks. In Doncieux, S. et al., editors, LNAI - Simulation of Adaptive Behavior, 6226, pp 435-446, Springer-Verlag, (2010). doi: 10.1007/978-3-642-15193-4_41.
[2022]
[2021]
[2020]
[2019]
[2018]
[2017]
[2016]
[2015]
[2014]
[2013]
[2012]
[2011]
Proceedings

MARIE SKŁODOWSKA-CURIE ACTIONS: Individual Fellowships (IF) “SpikeControl”
“Cerebellar Spiking Model For Real-time Closed-loop Sensorimotor Control”
Understanding how the brain processes and represents information is at the core of experimental studies of the Central Nervous System (CNS). A network of brain subsystems mediates information processing through distributed neural computation and dynamic patterns of neural activity. Over the last decades, studying how these patterns are elicited in the CNS under specific behavioural tasks has become a break through research topic in integrative neuroscience. These specific tasks are related to the concept of embodied cognition, according to which the primary goal of the CNS is to solve and facilitate the body-environment interaction. This project focuses on the cerebellum, a brain region that plays a crucial role in body-environment interaction, with a primary function related to adaptive motor control and coordination. The functional characteristics of the cerebellum make it a perfect candidate to start modelling and building an embodied nervous system. The cerebellar capability of performing adaptive information processing mediating sensorimotor control will be evaluated in specific tasks. Additionally, the emergence of cognitive-like representations will be studied by focusing on how models of the environment/tools can be acquired through a closed-loop sensorimotor interaction. This project sets forth a multidisciplinary methodology combining neuromimetic models and embodied neurorobotics. Simulated neural models and robotic experiments will guarantee full access to the system properties, which will be assessed through both qualitative and quantitative performance indicators to facilitate a constructive cross-validation against neurophysiological data. This approach will also allow us to predict new functional roles of specific cell/network/topology properties. The goal of this project lies on moving forward the knowledge frontiers in integrative neuroscience and biological control.
Description
Amongst computational models of various brain regions, the well-organized structure of cerebellum, has received special attention from researchers belonging to very different fields. On the one hand, neurophysiologists have studied and proposed detailed models and descriptions according to experimentally recorded cells and synaptic properties. However, these current detailed models are not meant to perform specific tasks at a behavioral or cognitive level (equivalent to awake animal protocols) as in functional experiments to control real agents (as front-end body). On the other hand, engineers have proposed machine-like systems that try to solve particular biological problems from a systemic point of view. Based on these opposed approaches, several cerebellar modeling frameworks have been proposed:
In state-generator models, the granule cell layer presents on/off type “granule” entities that provide a sparse coding of the state space (Marr-Albus Model(Marr, 1969;Albus, 1971) ,CMAC(Albus, 1975) model, or Yamazaki and Tanaka model(Yamazaki and Tanaka, 2005;2007a;2009;Yamazaki and Nagao, 2012)). These models succeed in explaining some traditional cerebellum-involving tasks such as eyelid conditioning(Yamazaki and Tanaka, 2007b) or motor control tasks(Manoonpong et al., 2007;Luque et al., 2011b;a). In functional models, only the functional abstraction of specific cerebellar operations are considered (MPFIM model(Wolpert and Kawato, 1998), Adaptive Filter model(Fujita, 1982;Porrill and Dean, 2007;Dean and Porril, 2008;Dean and Porrill, 2010;Dean et al., 2010), APG model(Houk et al., 1996), or LWPR model(Tolu et al., 2012)). This kind of approximation derives from an engineering point of view and can solve most of the tasks performed by common cerebellar models, such as eyelid conditioning, the vestibule ocular reflex (VOR), or movement correction. Finally, cellular-level models capture the biophysical features of cerebellar neuronal processing, and can be evaluated in the framework of neurophysiological experiments. These models are highly biologically plausible, but their application in the context of large-scale cerebellar modeling and computation remains limited. The very first approximations in this field were developed based on the Schweighofer–Arbib model(Schweighofer et al., 1998;Schweighofer et al., 1998b). Therefore, it can be noticed that there is a gap to be covered where neurophysiologists and system engineers need to work hand in hand towards a better understanding of how specific problems are solved using biologically-plausible computational principles(De Schutter, 2008).
As aforementioned, computational models of various brain regions have been developed and studied for more than thirty years in order to analyze central brain functions. Computational neuroscience (CN) is the natural complement of experimental brain research, since it provides a synthetic explanation about specific mechanisms and models which are only partially observed using anatomy, physiology or behavioral experimentation.
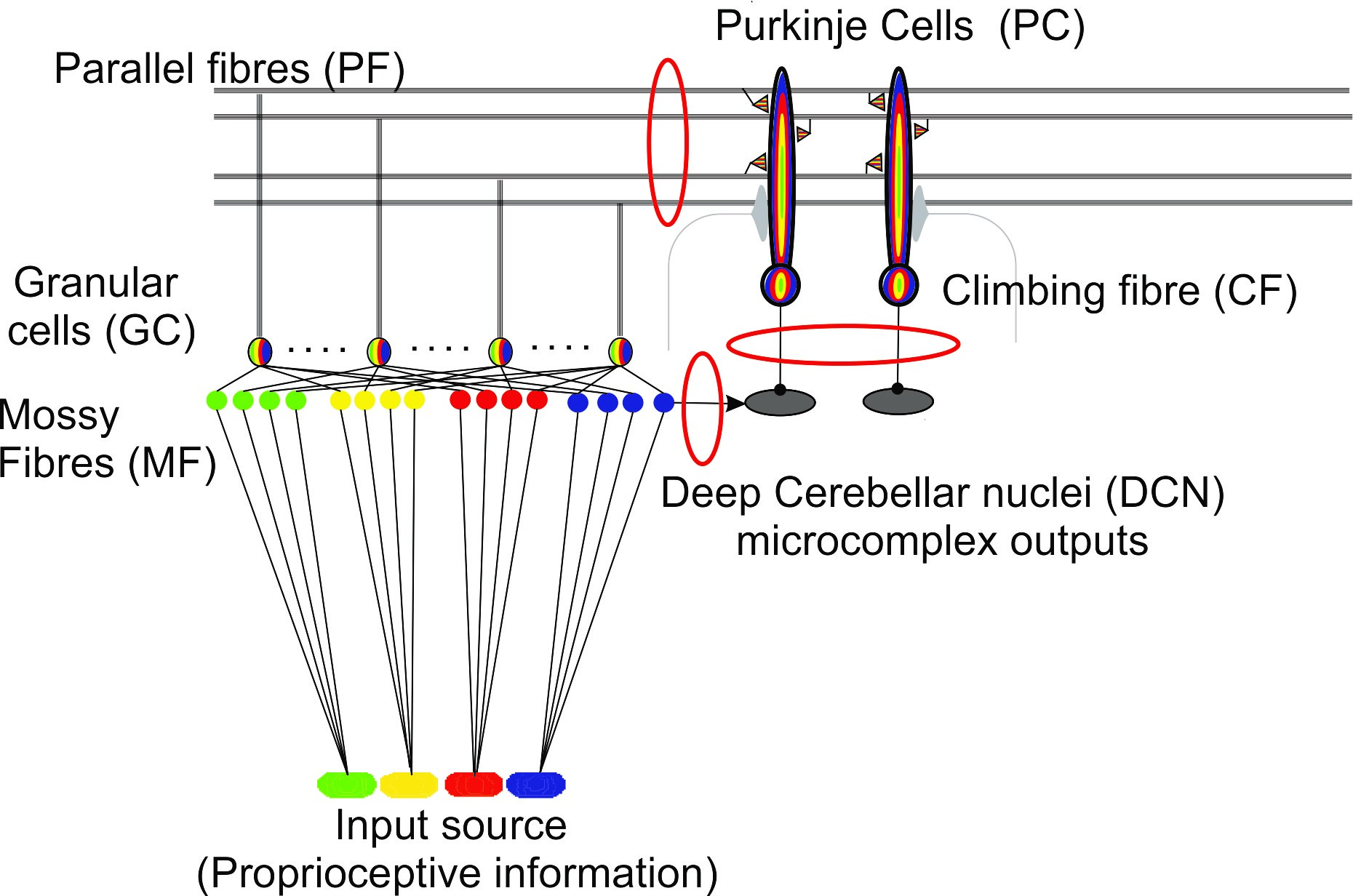
Fig. 1 Cerebellar microcircuitry. Distributed plasticity within the cerebellar microcircuitry is indicated by the red ovals.
It is a fact that, many computational models about the cerebro-cerebellar loop have been proposed since Marr and Albus thus providing elegant explanations about the core of the forward controller operation that cerebro-cerebellar loop seems to carry out. Nevertheless, these computational theories tend to focus in one part of the cerebellar circuitry and then extrapolating the obtained conclusions to the whole cerebro-cerebellar system. It is also true that functional features are not either suddenly going to emerge from modeling all the cerebellar parts together since small deviations in many of the estimated network parameters (Sporns, 2006) can cause large deviations in resultant global behavior. But, it is also true that simulating nervous systems "connected" to a body (agent or robot with sensors and actuators) could be of great interest for studying how certain capabilities of the nervous system (e.g. the role of the cerebellum in coordinated movements and object manipulation) are based on cellular characteristics, nervous system topology or local synaptic adaptation mechanisms. This project represents an integrative approach which builds the bridge between task specific experimentation (equivalent to “awake animal experimentation”) and Systems Neuroscience models.
This allows studying the role of certain nervous systems under what it is called "behavioral/cognitive tasks". This is closely related to the concept of "embodied cognition" (Pfeifer and Bongard, 2007), in which the main aim of the CNS is to solve and facilitate the body interaction with the environment. Therefore, it is crucial to study nervous system models within the framework of its interaction with a body (sensors and actuators) and environment.
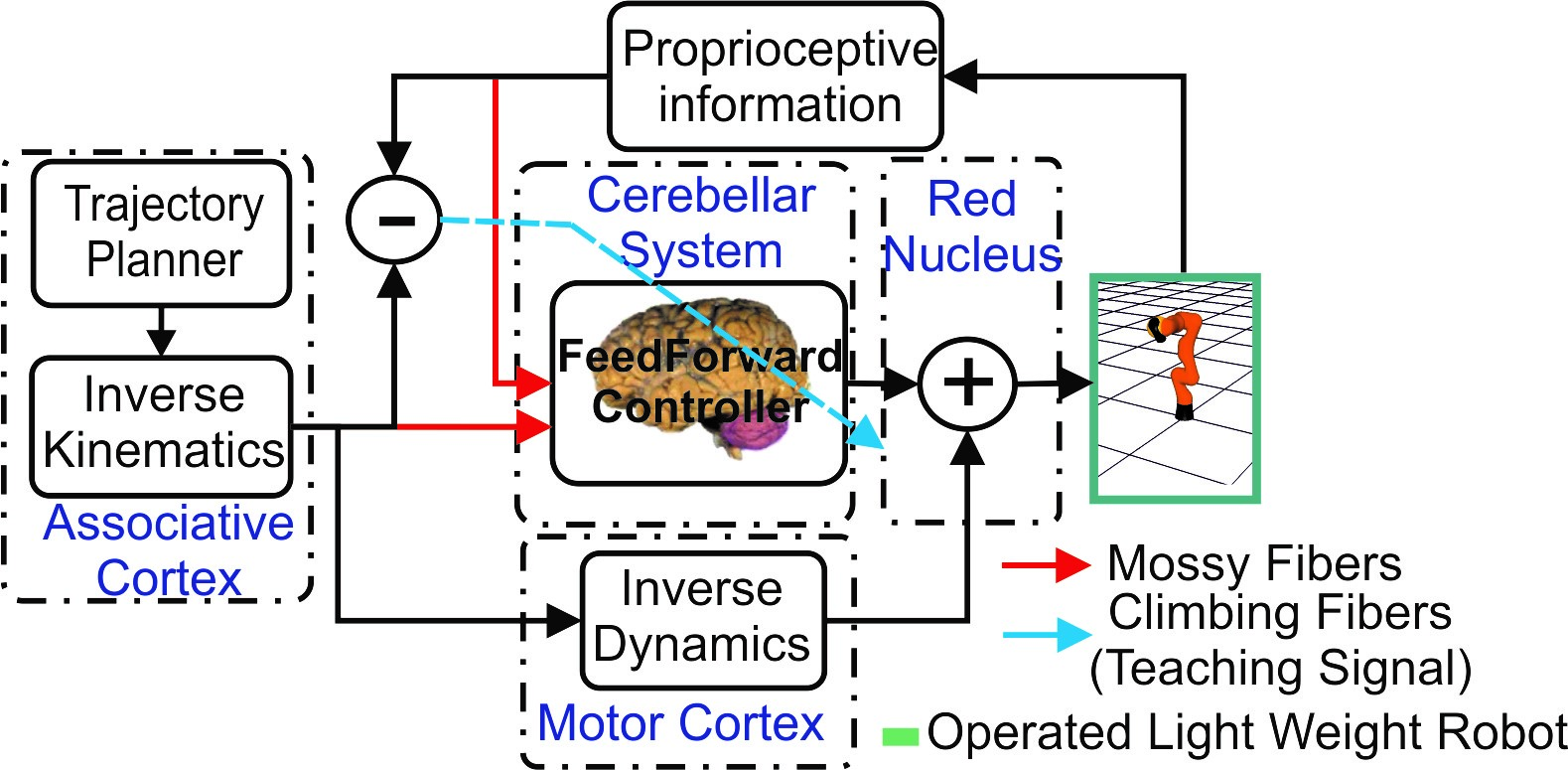
Fig. 2. Cerebellar control loop. The cerebellum provides corrective commands to compensate the mismatch between the inverse dynamic model and the real robot.
The present project focuses on an integrated approach to the cerebellar circuit modeling within real time “behavioral and cognitive tasks”. This is a compromise approach based on the assumption that most cerebellar functions involve the cooperative computation of several neural subcircuits and circuits
References
Albus, J.S. (1971). A theory of cerebellar function. Math Biosci. 10, 25-61.
Albus, J.S. (1975). Data storage in the cerebellar model articulation controller (CMAC). J. Dyn. Syst. Meas. Contr. ASME 3, 228–233.
De Schutter, E. (2008). Why Are Computational Neuroscience and Systems Biology So Separate? PLoS Comput Biol 4, e1000078.
Dean, P., and Porril, J. (2008). Adaptive filter Models of the Cerebellum. Computational Analysis. The Cerebellum 7, 567-571.
Dean, P., and Porrill, J. (2010). The cerebellum as an adaptive filter: a general model? Funct. Neurol. 25, 173-180.
Dean, P., Porrill, J., Ekerot, C.F., and Jörntel, H. (2010). The cerebellar microcircuit as an adaptive filter: experimental and computational evidence. Nat. Rev. Neurosci. 11, 30-34.
Fujita, M. (1982). Adaptive filter model of the cerebellum. Biol. Cybern. 45, 195-206.
Houk, J.C., Buckingham, J.T., and Barto, A.G. (1996). Models of cerebellum and motor learning. Behavioral and Brain Sciences 19, 369-383.
Luque, N.R., Garrido, J.A., Carrillo, R.R., Coenen, O.J.M.D., and Ros, E. (2011a). Cerebellar Input Configuration Toward Object Model Abstraction in Manipulation Tasks. Neural Networks, IEEE Trans. 22, 1321-1328.
Luque, N.R., Garrido, J.A., Carrillo, R.R., Coenen, O.J.M.D., and Ros, E. (2011b). Cerebellarlike Corrective Model Inference Engine for Manipulation Tasks. Systems, Man, and Cybernetics, Part B: Cybernetics, IEEE Trans. 41, 1299-1312.
Manoonpong, P., Geng, T., Kulvicius, T., Porr, B., and Wörgötter, F. (2007). Adaptive, Fast Walking in a Biped Robot under Neuronal Control and Learning. PLoS Comput Biol 3, e134.
Marr, D. (1969). A theory of cerebellar cortex. J. Physiol. 202, 437-470.
Pfeifer, R., and Bongard, J. (2007). How the body shapes the way we think - a new view on intelligence.
Porrill, J., and Dean, P. (2007). Cerebellar Motor Learning: When Is Cortical Plasticity Not Enough? PLoS Comput Biol 3, e197.
Schweighofer, N., Arbib, M.A., and Kawato, M. (1998). Role of the cerebellum in reaching movements in human. I. Distributed Inverse dynamics control. European J. Neurosci. 10, 86-94.
Schweighofer, N., Spoelstra, J., Arbib, M.A., and Kawato, M. (1998b). Role of the cerebellum in reaching movements in human. II. A neural model of the intermediate cerebellum. Eur.J. Neurosci. 10, 95-105.
Sporns, O. (2006). Small-world connectivity, motif composition, and complexity of fractal neuronal connections. Dedicated to the Memory of Ray Paton 85, 55-64.
Tolu, S., Vanegas, M., Luque, N.R., Garrido, J.A., and Ros, E. (2012). Bio-inspired Adaptive Feedback Error Learning Architecture for Motor Control. Biol. Cybern. 106, 507-522.
Wolpert, W., and Kawato, M. (1998). Multiple paired forward and inverse models for motor control. Neural Networks 11, 1317-1329.
Yamazaki, T., and Nagao, S. (2012). A Computational Mechanism for Unified Gain and Timing Control in the Cerebellum. PLoS ONE 3, e33319.
Yamazaki, T., and Tanaka, S. (2005). Neural modeling of an internal clock. Neural Comp. 17, 1032-1058.
Yamazaki, T., and Tanaka, S. (2007a). The cerebellum as a liquid state machine. Neural Networks 20, 290-297.
Yamazaki, T., and Tanaka, S. (2007b). A spiking network model for passage-of-time representation in the cerebellum. Eur. J. Neurosci. 26, 2279-2292.
Yamazaki, T., and Tanaka, S. (2009). Computational models of timing mechanisms in the cerebellar granular layer. Cerebellum 8, 423-432.

contact
Hopefully any questions you had about me will have been answered clearly now you've looked at this site. If there are still any unanswered questions, please feel free to use the contact form below..
- Download My CV PDF
- Niceto R. Luque Sola
- Research Centre for Information and Communications Technologies ,University of Granada
- Address: Calle Periodista Rafael Gómez Montero, 2, Office D 1.2 Postal Code: ES-18014 Granada, Spain
- Phone : +34 958 24 17 75
- Email : nicetoluque@gmail.com
